
FDA issues a warning letter to Megafine Pharma Limited for data integrity violations.

FDA issues a warning letter to Megafine Pharma Limited for data integrity violations.

Samsung Bioepis and partner Biogen announced on May 30 that the European Commission approved Flixabi for the treatment of six inflammatory conditions.

The agency cited Tai Heng Industry with CGMP deviations, including failure to ensure data integrity.

Sandoz is seeking approval for the same indications as Roche’s reference product MabThera.

FDA accepted for review Samsung Bioepis’ BLA for SB2, a biosimilar to Remicade (infliximab).

The company recalls products due to sterility concerns.

The agency detailed its 2015 achievements in an annual report.

The agency provides guidelines on generating histopathology data in nonclinical biomarker qualification studies.

FDA is advising there may be serious and potentially permanent side effects associated with taking fluoroquinolone antibacterial drugs consistently.

Biomedical innovation legislation may stall in 2016.

The updated guidelines detail best practices for conducting research with human embryonic stem cells.

Drug manufacturing lapses undermine NIH research programs.

The agency and national authority representatives talk about regulators’ role in keeping drug prices affordable.

The agency summarizes the results of its 10-year-long initiative to support small- and medium-sized enterprises.

Members of ABPI, GSK, AstraZeneca, and the BioIndustry Association composed a letter explaining why the UK remaining part of the EU benefits the life-sciences sector.

Phase I clinical trials of PfSPZ revealed it may protect healthy adults, who have not been exposed to Malaria before, for more than on year.

The agency cited Apotheca Supply, a repackager and relabeler of pharmaceuticals for CGMP violations.

Progress is being made, but work remains to be done. Brian Daleiden, vice president of marketing at TraceLink, discusses the state of the industry, as companies attempt to meet complex, and changing, global requirements.

A federal judge entered an order of permanent injunction against Paul W. Franck, the owner and operator of several compounding pharmacies in Florida.

Siegfried Schmitt, Principal Consultant, PAREXEL International, discusses how to report quality metrics to FDA.
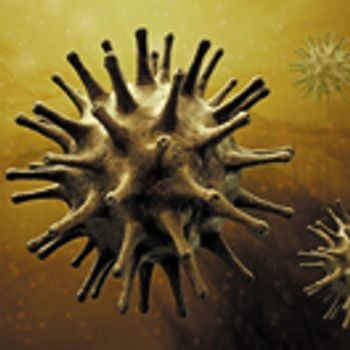
Testing product and process intermediates alone is helpful, but does not provide a complete solution to viral safety. This article proposes integrated solutions for systemic and proactive viral risk mitigation.

Immuno-oncology drugs are demonstrating patient benefits, but growing resistant to the high cost has implications for patients, market access, and manufacturers.

The agency extends the indication of the drug in combination with bendamustine.

The agency holds a workshop to strengthen collaboration with healthcare providers.

The company voluntarily recalls product due to particulate matter.