
Webcasts
Webinar Date/Time: Tue, Jun 23, 2026 11:00 AM EDT

Webcasts
Webinar Date/Time: Tue, Jun 23, 2026 11:00 AM EDT

Sponsored
CDMOs are changing tactics in order to meet faster timelines, manage multiple manufacturing platforms, and navigate the industry's shift toward complex biologics.

Webcasts
Webinar Date/Time: Thu, Apr 30, 2026 11:00 AM EDT

Webcasts
Webinar Date/Time: Wed, Oct 22, 2025 11:00 AM EDT

Webcasts
Webinar Date/Time: Wed, Sep 24, 2025 11:00 AM EDT

Webcasts
Tue, Jul 15, 2025 11:00 AM EDT

Webcasts
Webinar Date/Time: Thu, May 15, 2025 11:00 AM EDT

Webcasts
Webinar Date/Time: Thursday, December 5th, 2024 at 11:00 AM EST

Sponsored Content
Key strategies for accelerating development and manufacturing and development timelines to bring drugs to market faster and give developers a competitive advantage.

Webcasts
Webinar Date/Time: Wed, Mar 20, 2024 11:00 AM EDT

Webcasts
Webinar Date/Time: Thu, Feb 22, 2024 11:00 AM EST

Webcasts
Webinar Date/Time: Wed, Dec 13, 2023 11:00 AM EST

Sponsored Content
Custom Protein Production at the Highest Levels of Quality and Speed

Sponsored Content
WuXi biologics provides end-to-end biologics and vaccines solutions.
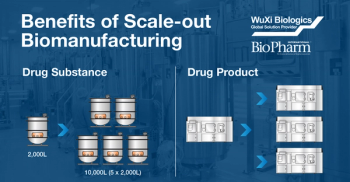
Sponsored Podcast
Changes in production scale required due to increasing product needs throughout clinical trials introduces risk to the product development timeline. Both the process and the product can be dramatically impacted from scaling-up to larger production volumes. Once in commercial manufacturing, sudden changes to product demand in either direction can have costly consequences if the scale of production chosen is not adequate to meet the changing material requirements. Adapting a “scale-out” instead of “scale-up” strategy for clinical and commercial production can greatly minimize these risks. This podcast will review the scale-out biomanufacturing approach and the significant benefits of this production paradigm.

Sponsored Content
WuXi biologics provides end-to-end biologics and vaccines solutions.

Sponsored Content


Webcast
***Live: Thursday, Dec. 3, 2020 at 11am EST| 8am PST| 4pm GMT| 5pm CET*** Review preclinical strategies to improve drug development outcomes for biologic drugs, including innovative solutions and risk-based approach to expedite investigational new drug submissions, especially for pandemic response initiatives.. ***On demand available after final airing until Dec. 3, 2021***

March 1st 2021

October 18th 2021