Transcriptomics plays a role in influencing the production of recombinant therapeutics in microbial and mammalian hosts.

Transcriptomics plays a role in influencing the production of recombinant therapeutics in microbial and mammalian hosts.
The National Institute of Standards and Technology (NIST) has developed Standard Reference Material (SRM) 2082 as a pathlength standard for UV absorbance measurements for use with the new generation of microvolume spectrophotometers and short-pathlength cuvettes.
Process understanding and careful assessment of risks are essential in developing viral clearance programs.

Geofrey Wyatt has been named CEO and Clifford Wyatt named president of Wyatt Technology.

The company’s investment in new laboratory equipment and personnel will strengthen its cell line development services.

The company showcased newer products and equipment for cell culture laboratories at Analytica 2018 in Munich, Germany.

The new platform is expected to speed up cell line development.

A new scientific publication examines analytical processes for the emerging legal cannabis industry.

RGtimeline/Shutterstock.comParenteral product quality is improving.

Access to multiple analytical techniques is essential for fully characterizing complex protein formulations.

The Concentrating Pipette Select from InnovaPrep is a small automated benchtop instrument that can concentrate contaminates from large volumes of liquids for improved detection.
Outsourcing analytics can be a cost-effective way for biopharma companies to adapt to new technologies and regulations.

The company’s close communication with customers has enabled it to bring advanced pipetting products to market.

The company’s Krios G3i cryo-electron microscope is a finalist in the 2018 Edison Awards.

Gyros Protein Technologies’ new immunoassay technology includes expanded software to optimize time to results and sample capacity while simplifying workflows.

Telstar reports that its Boreas, a new 86° ultra-low-temperature freezer, increases average performance by 20%.

Contract testing organizations can provide bio/pharma companies with a cost-effective way to adapt to new technologies and regulations.
Successful outsourcing relationships for early phase analytics in drug development are driven by partnership.

Revisions to chapters on glass containers and elastomeric closures were canceled following review of comments.

Fluid Imaging has introduced a new imaging flow microscope at Pittcon 2018 in Orlando, FL.

LabVantage offers laboratory information management system solutions that include additional industry-specific functionalities.

CEM reports that the EDGE extraction system for rapid sample preparation of gas chromatography/liquid chromatography samples from CEM can prepare samples for molecular analysis within 5 min.

The company showcased various updated and new analytical systems and solutions for food analysis and pharmaceutical, clinical research, and materials sciences applications.

New, user-friendly analytical systems provide high-throughput comprehensive characterization and quantitation of small and large molecules.
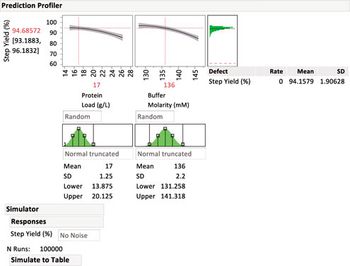
Statistical methods to identify critical process parameters and critical material attributes-and approaches to control them-are needed to protect drug product and drug substances.