
Increased capacity will help Circio progress development of circVec AAV gene therapy and in-vivo cell therapy, with the company setting a goal of selecting its first therapeutic candidate in 2026.
Patrick Lavery is an Editor for the MJH Life Sciences brands Pharmaceutical Technology and BioPharm International, and their respective websites, pharmtech.com and biopharminternational.com. Previously at MJH, he filled the same role for LCGC International and Spectroscopy.

Increased capacity will help Circio progress development of circVec AAV gene therapy and in-vivo cell therapy, with the company setting a goal of selecting its first therapeutic candidate in 2026.
Biocon said that, according to clinical data, both Vevzuo and Evfraxy demonstrated a safety and efficacy profile comparable to that of the reference product.
Linvoseltamab-gcpt, marketed as Lynozyfic, is a bispecific antibody designed to facilitate T cell activation and cancer cell killing.

The expansion of the Kentucky site accounts for $80 million of the announced investment, with the remaining money earmarked for facilities in Michigan.
The new building will create 150 new jobs and expands total production area to nearly 12,000 square meters.
Using its proprietary Optimer delivery vehicle, the company said it uncovered a previously unexploited protein marker expressed on activated hepatic stellate cells.

In contrast to earlier conferences in 2025, experts interviewed as part of the BIO conference in Boston did not come to a consensus about lasting impacts of recent changes in US government policy.

In preclinical trials in mice, the vaccine delivery system provided protection against malaria that was strong enough to stand up to the efficacy of traditional multi-dose regimens.

A key property included in the acquisition is farabursen, an investigational next-generation oligonucleotide targeting the microRNA miR-17 with preferential kidney exposure, intended to treat people with autosomal dominant polycystic kidney disease.
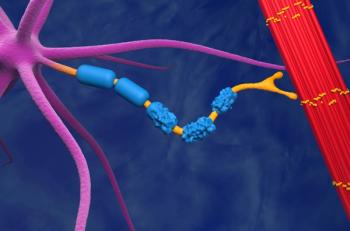
Amyotrophic lateral sclerosis, or Lou Gehrig’s disease, is the most common form of motor neurone disease (MND), with more than 5000 people in the United States being diagnosed with MND annually.

Matthew Stober and Campbell Bunce of Abzena talk about the CDMO’s role in accelerating the path of newer and more effective modalities to market.

The department’s newly announced partnerships were part of a weeklong visit that included discussions held at BIO 2025.

Some of the companies that received American samples, FDA said, are partially owned or controlled by the Communist Party in China.

FUJIFILM representatives talked about the rebrand of the company’s life sciences businesses, partnerships with Regeneron and other companies, and the importance of having a presence at conferences like BIO 2025.

Bartłomiej Czubek, director of Business Development at Mabion, discussed what’s working, and the challenges set forth by what isn’t working, in the relationships built within the present biologics CDMO market.

Korro Bio CEO and president Ram Aiyar, PhD, delves into his company’s partnership with Novo Nordisk as a means of reaching a larger patient population with new ideas and approaches.

Korro Bio CEO Ram Aiyar discusses his company’s platform that tunes the RNA editing process to target a wider range of rare diseases.

The company’s successful completion of facility and equipment qualifications gives it another biologics manufacturing option alongside its United States campus in Morgan Hill, Calif.

The collaboration will focus on identifying pre-clinical candidates for high-priority targets, including a small molecule oral therapy for immunological diseases.

The agreement, which covers a period of five years, has been designed to offer a turnkey service to companies seeking faster time to market for ADCs and bioconjugates.

The partnership leverages the Hesperos organ-on-a-chip platform in the preclinical development of Psilera’s lead compound targeting the progressive neurological disorder for which treatment options are few.

The facility is on the company’s Bedford, NH campus, where another existing building will be repurposed into a Development Center of Excellence.

Quality, stability, sustainability, and the increased and thoughtful integration of artificial intelligence are foremost in the minds of those on the leading edge of testing trends.

TriLink’s CleanCap capping technology, to which Quantoom will have access in the new partnership, has been used in the majority of all approved COVID-19 mRNA and saRNA vaccines.

Nicholas Giovannone of Regeneron explained the strategies behind overcoming re-administration challenges in a recap of his presentation at ASGCT 2025.
In this recap of a poster presentation, Austin Daniels of Yokogawa Fluid Imaging Technologies explains how flow imaging microscopy can detect the amount and types of both subvisible and visible particles in samples not often detected by traditional methods.

Jerry Chapman of Redica Systems spoke with BioPharm International® about how artificial intelligence can assist in analyzing a clinical site to determine risk factors and spotlight potential future risks.

In discussions at INTERPHEX 2025, company representatives seized upon several common themes, most notably safety, sustainability, and the increasing utility of automation.

Biocon Biologics’ Yesintek demonstrated comparable safety and efficacy to the originator project, according to clinical data from a trial program.

The company’s presence at ASGCT included not only a booth on the show floor, but also one oral and nine poster presentations.