
Developing a quality agreement template for single-use systems.

Developing a quality agreement template for single-use systems.

US Pharmacopeia promotes horizontal standards and a product-class approach for quality attributes.

Rising imports, overseas production spur collaboration and realignment of enforcement activities.
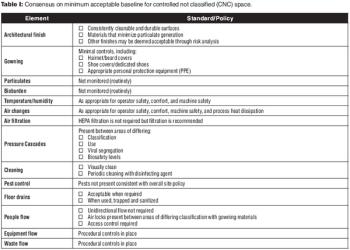
The authors re-examine environmental controls in the context of technical advances in manufacturing.

Incorporating regulatory requirements into the product life cycle is crucial.

Follow-on versions of complex biologics require extensive expertise.
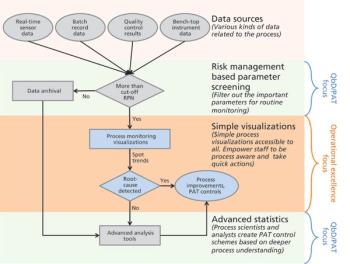
The authors focus on operational excellence in manufacturing of biotechnology therapeutic products in the QbD paradigm.

Industry struggles to curb drug abuse, diversion, and disruptions in supply to ensure access to quality products.
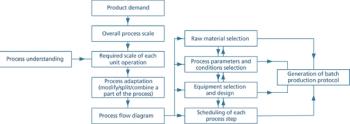
Clear documentation and open communication are essential for effective technology transfer.

A rigorous cost-benefit assessment can help to chart a cost-effective path forward.

FDA, NIH, and industry seek new strategies to spur drug development and promote access to therapies.

Courts and Congress seek to reshape policies and programs affecting drug costs and access.

New supply-chain challenges are forcing companies to act in different ways to secure product safety.

Executive management leadership is essential in the effective implementation of QbD.

US Pharmacopeia develops and improves its class approach for ensuring quality biopharmaceuticals.
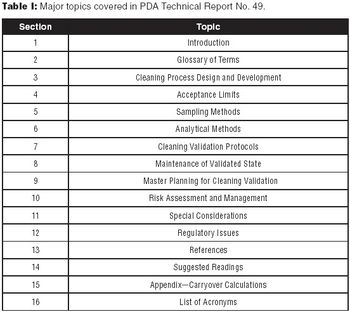
The authors encourage biotech manufacturers to consult PDA Technical Report No. 49 for a detailed perspective on current practices and issues in biotech cleaning validation.

As drug shortages make headlines, FDA tests the Sentinel safety system and its effect on healthcare.

FDA prepares for top-level changes while promoting transparency and product safety

An approach to reduce batch time, increase productivity, and decrease costs.

Top priorities for manufacturers include user fees, new health initiatives, and regulatory compliance.

Best practices to strengthen supplier quality management.

The pathway for biosimilar approval in the US has been set. But are US patients too far behind Europe?

Comparative effectiveness poses challenges for drug manufacturers.

A new strategy to streamline vaccine development and oversight.

Industry and regulators disagree over noncritical parameters.